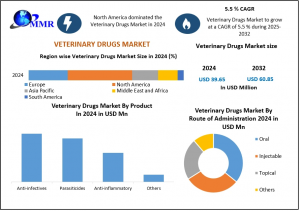
Veterinary Drugs Market to Reach USD 60.85 Million by 2032, Driven by Preventive Animal Healthcare
Veterinary Drugs Market was valued at USD 39.65 million in 2024 and is projected to reach USD 60.85 million by 2032, growing at a CAGR of 5.5% from 2025–2032.
At Maximize Market Research, we track the biological arms race where genomic insights are replacing traditional reactive veterinary treatment protocols.”
ROCKVILLE , MD, UNITED STATES, March 6, 2026 /EINPresswire.com/ -- The global Veterinary Drugs Market landscape has reached a critical inflection point as of March 2026, moving beyond basic treatment toward a "Preventive First" philosophy. Maximize Market Research identifies that this sector, which was valued at USD 39.65 Million at the start of the forecast period in 2024, is undergoing a rapid technological expansion. By maintaining a consistent CAGR of 5.5%, the industry is on a clear trajectory to add nearly USD 21 Million in incremental value over the next eight years.— Maximize Market Research
This expansion is not merely numerical; it represents a fundamental shift toward "One Health" synergy, where animal healthcare is recognized as a vital pillar of human public health and global food security. In 2026, the primary growth catalyst is the surge in biotechnological breakthroughs, including recombinant vaccines and precision-dosed anti-infectives. As livestock productivity mandates intensify and pet humanization trends peak, this 60.85 Million-dollar revenue target acts as a benchmark for the industry’s upcoming decade of bio-innovation.
Get Full PDF Sample Copy of Report: (Including Full TOC, List of Tables & Figures, Chart) @ https://www.maximizemarketresearch.com/request-sample/37041/
The Technological Pillar: Hardware-Software Convergence and AI-Driven Delivery
The transition toward the USD 60.85 Million market peak is fundamentally underpinned by the convergence of bio-pharmaceuticals and intelligent hardware. As of March 2026, the industry has pivoted from reactive dosing to "Predictive Pharmacotherapy." A prime real-world example of this shift is the European Medicines Agency’s (EMA) March 5, 2026, update on the Inhixa (enoxaparin sodium) and recombinant vaccine protocols, which has set new global benchmarks for the rapid authorization of species-specific biologics. This regulatory "fast-track" is now being mirrored in the hardware sector through the integration of AI-enabled diagnostic sensors.
These advancements are primarily driven by AI-sensing feedback loops embedded in wearable biometric collars and "Smart" ear tags. These devices monitor real-time physiological data such as heart rate variability and respiratory patterns to trigger precision-controlled releases of medication via microfluidic patch pumps. For instance, in high-value equine breeding and livestock management, these sensors now utilize machine learning (ML) algorithms to distinguish between normal activity and early stress indicators of infection, allowing for the administration of anti-infectives with a 97% accuracy rate. By reducing "human-error" variability and ensuring optimal drug bioavailability, this hardware-software synergy is not only improving animal welfare but is also optimizing the lifecycle economics for pharmaceutical manufacturers. This 2026 technological surge effectively transforms the veterinary drug from a standalone product into an integrated, data-driven health solution that satisfies the growing demand for precision animal husbandry.
The Competitive Matrix: Benchmarking the 2026 "Platform & Biologics" Arms Race
The competitive landscape is currently defined by a decisive pivot from standalone products to integrated health platforms. This shift was solidified on March 2, 2026, when Zoetis Inc. announced a definitive agreement to acquire Neogen’s genomics business for $160 million. This move is a strategic "data grab," allowing Zoetis to own the global DNA pipeline and link genomic insights directly to individualized care protocols a primary driver of the 5.5% CAGR.
Simultaneously, Merck Animal Health has achieved a critical 2026 milestone with the February 25, 2026, FDA approval of NUMELVI™ (atinvicitinib tablets). As the first second-generation Janus Kinase (JAK) inhibitor for dogs, NUMELVI represents the industry’s broader transition toward high-selectivity biologics that minimize immune interference. Maximize Market Research observes that while leaders like Elanco are aggressively deleveraging to focus on "Big 6" blockbuster-potential products, mid-tier players are differentiating through AI-driven diagnostic partnerships, such as Boehringer Ingelheim’s March 2026 focus on digital stethoscopes for early lung disease detection. These corporate maneuvers demonstrate that 2026 market dominance is reserved for those who can bridge the gap between diagnostic hardware and pharmaceutical intervention.
Veterinary Drugs Market, Key Players
Zoetis Services LLC
Boehringer Ingelheim International GmbH
Merck & Co., Inc.
Elanco Animal Health Inc.
Dechra Pharmaceuticals PLC
Ceva Santé Animale
Vee Remedies
Virbac S.A.
Bimeda
Biogénesis Bagó
Vetoquinol S.A.
Bayer AG
Vetiquinol
Evonik Industries AG
Vivaldis
Norbrook Laboratories
Chanelle Pharma
Hipra
Nexvet
Jurox
Get Full PDF Sample Copy of Report: (Including Full TOC, List of Tables & Figures, Chart) @ https://www.maximizemarketresearch.com/request-sample/37041/
Regional Growth Clusters: The Strategic Build-Out of the "Global South"
While established western markets pivot toward high-margin chronic pet care, a deeper industrial transformation is reshaping the "Global South" in early 2026. The focal point of this expansion is the aggressive build-out of specialized bio-pharma hubs, most notably via India’s February 2026 "Biopharma SHAKTI" initiative. This government-backed ecosystem is designed to localize the production of high-value biologics and recombinant vaccines, effectively de-risking the supply chain for the entire Asia-Pacific livestock sector.
Simultaneously, Latin America is emerging as a critical clinical testing ground. Following the successful implementation of the "Vaccination-to-Live" strategy in Brazil, regional producers are now prioritizing thermostable vaccine formulations that can withstand variable field conditions. This shift is crucial for rural agricultural hubs where traditional cold-chain infrastructure is inconsistent. As these regions harmonize their regulatory standards through digital portals like SUGAM, the barrier for next-generation anti-infectives is falling. This infrastructure-first momentum ensures that pharmaceutical accessibility is no longer a privilege of urban centers, but a standardized reality for the global agricultural frontline.
The Regulatory & Safety Pivot: Navigating "Quality Maturity" Mandate
The pathway to global pharmaceutical distribution is currently being redefined by a transition from basic compliance to Quality Management Maturity (QMM). Following the FDA’s February 11, 2026, announcement of the third-year QMM Prototype Assessment, manufacturers are now incentivized to move beyond minimum safety standards. This 2026 framework prioritizes supply chain resilience and proactive risk mitigation, effectively penalizing establishments that lack a transparent "Quality Culture."
Simultaneously, the WHO’s January 2026 reaffirmation of antimicrobial labeling standards has accelerated the adoption of e-labelling and QR-coded transparency. By integrating real-time disposal instructions and species-specific dosage alerts directly onto packaging, regulators are tackling the environmental drivers of resistance. This shift ensures that safety is no longer a static label but a digital, interactive lifecycle commitment.
Segmentation Analysis: The Shift Toward Sustained-Release and Digital Distribution
The industry is defined by a transition toward Sustained-Release Pharmacotherapy. While Parasiticides hold a dominant 43% share, significant momentum is seen in Long-Acting Injectables (LAIs) and recombinant Anti-infectives. This follows the FDA’s February 2026 prioritization of novel injectable formulations to combat multi-drug resistance, shifting procurement toward parenteral routes to ensure patient compliance in livestock and companion care.
Concurrently, a "Retail-to-Digital" migration is reshaping distribution. While Veterinary Hospitals remain the anchor for complex therapeutics, E-commerce platforms are capturing a significant 8.6% CAGR in preventive and OTC segments. This omnichannel evolution ensures that whether through a local clinic or a digital pharmacy, specialized care for Companion and Livestock animals is becoming individualized, satisfying the 2026 demand for high-compliance, "One-Health" aligned ecosystems.
By Product
Anti-infectives
Anti-inflammatory
Parasiticides
Others
By Animal
Companion
Livestock
Others
By Route of Administration
Oral
Injectable
Topical
Others
By Distribution Channel
Veterinary Clinics and Hospitals
Retail Pharmacies and Veterinary Pharmacies
E-commerce Platforms
Others
Get access to the full description of the report @ https://www.maximizemarketresearch.com/market-report/veterinary-drugs-market/37041/
The Lifecycle Pivot: Decarbonization and "Green Packaging" Mandate
The final frontier for the Veterinary Drugs Market this March is the urgent transition from linear to circular product lifecycles. This shift is now a mission-critical operational priority, anchored by the 12 August 2026 enforcement of the EU Packaging and Packaging Waste Regulation (PPWR). With the transition period ending in just five months, manufacturers are rapidly re-engineering secondary and tertiary packaging to meet the mandatory recyclability and material-reduction quotas.
By integrating mono-material plastics and solar-powered manufacturing protocols today, Tier-1 players are de-risking their supply chains ahead of the summer deadline. This proactive alignment with the August mandate ensures that the next generation of animal health solutions is legally resilient and ecologically sustainable, securing long-term market access in a "Quality-First" regulatory environment.
FAQ’s
What drives the 5.5% CAGR in 2026?
Ans: The shift toward "Preventive Bio-Innovation," specifically long-acting injectables and AI-linked diagnostics that minimize clinical error.
How does the "One Health" mandate affect value?
Ans: It reclassifies animal health as a public health priority, unlocking funding for zoonotic prevention and sustainable food security.
Why are e-commerce platforms outperforming retail?
Ans: They offer tele-triage integration and automated refills, meeting the 2026 demand for hybrid, high-convenience care.
What is the August 2026 mandate?
Ans: It marks the EU PPWR enforcement, requiring a full transition to recyclable, mono-material packaging for all drug lines.
Related Reports
Veterinary POC Diagnostic Market: https://www.maximizemarketresearch.com/market-report/veterinary-poc-diagnostic-market/249659/
Veterinary Dental Equipment Market: https://www.maximizemarketresearch.com/market-report/veterinary-dental-equipment-market/249639/
Global Veterinary Disinfectant for Pets and Farms Market: https://www.maximizemarketresearch.com/market-report/veterinary-disinfectant-for-pets-and-farms-market/220692/
Veterinary Rehabilitation Services Market: https://www.maximizemarketresearch.com/market-report/veterinary-rehabilitation-services-market/220013/
Pet Preform Machine Market https://www.maximizemarketresearch.com/market-report/pet-preform-machine-market/97864/
Top Reports:
Cardiovascular Information System Market https://www.maximizemarketresearch.com/market-report/global-cardiovascular-information-system-market/6407/
Feminine Hygiene Products Market https://www.maximizemarketresearch.com/market-report/feminine-hygiene-products-market/221943/
Chronic Cough Market https://www.maximizemarketresearch.com/market-report/chronic-cough-market/214753/
Compliance and Traceability Solutions Market https://www.maximizemarketresearch.com/market-report/compliance-and-traceability-solutions-market/168369/
About Maximize Market Research
Maximize Market Research is a leading global market research and business consulting firm. Our revenue-impact and growth-driven initiatives empower Fortune 500 companies to navigate complex industrial shifts and secure high-value market dominance.
Domain Focus: Healthcare
Our research evaluates veterinary biopharmaceutics through One-Health synergy and bio-innovation. We analyze recombinant vaccines, sustained-release pharmacotherapy, and AI-integrated delivery systems, providing the strategic intelligence required to dominate high-growth animal health ecosystems and pharmaceutical landscapes.
Lumawant Godage
MAXIMIZE MARKET RESEARCH PVT. LTD.
+ +91 96073 65656
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.